 It is important to recognize which numbering system is being used and to be able to find the number of valence electrons in the main block elements regardless of which numbering system is being used. You will come across periodic tables with both numbering systems. In this numbering system, group 1A is group 1 group 2A is group 2 the halogens (7A) are group 17 and the noble gases (8A) are group 18. The elements in this group are also gases at room temperature.Īn alternate numbering system numbers all of the \(s\), \(p\), and \(d\) block elements from 1-18. We will learn the reason for this later, when we discuss how compounds form. Period - reactivity increases as you go from the left to the right. These elements also have similar properties to each other, the most significant property being that they are extremely unreactive, rarely forming compounds. Group - reactivity increases as you go down a group. Each INB activity is designed to help students compartmentalize information for a greater. The students will also be interacting with their journals using INB templates for periodic table and reactivity. This group contains very reactive nonmetal elements. The periodic table and reactivity lesson plan includes a PowerPoint with activities scattered throughout to keep the students engaged. Group 7A (or 17) elements are also called halogens. It is important to recognize a couple of other important groups on the periodic table by their group name. Remember, Mendeleev arranged the table so that elements with the most similar properties were in the same group on the periodic table. Following properties of elements show a very clear periodic trend in the periodic table. The same pattern is true of other groups on the periodic table. Once again, because of their similarities in electron configurations, these elements have similar properties to each other. Group 2A is also called the alkaline earth metals. Although most metals tend to be very hard, these metals are actually soft and can be easily cut. Trends in reactivity in the periodic table Resource RSC Education Resources Chemistry for the gifted and talented: trends in reactivity in the periodic table Bookmark This could be used to follow up some work on the periodic table where the trends in reactivity in groups 1 and 7 have been identified. These trends can be predicted merely by examing the periodic table and can be explained and understood by analyzing the electron configurations of the elements. Group 1A is also known as the alkali metals. The periodic table arranges the elements by periodic properties, which are recurring trends in physical and chemical characteristics. Because of their similarities in their chemical properties, Mendeleev put these elements into the same group. Four major factors affect reactivity of metals: nuclear charge, atomic radius, shielding effect and sublevel arrangement (of electrons). The elements within the same group of the periodic table tend to exhibit similar physical and chemical properties. The elements in 1A are all very reactive and form compounds in the same ratios with similar properties with other elements. 8.9: Examples of Periodic Chemical Behavior. This is what causes these elements to react in the same ways as the other members of the family. All of the 1A elements have one valence electron. A group is a vertical column of the periodic table. Remember that Mendeleev arranged the periodic table so that elements with the most similar properties were placed in the same group. A more sophisticated calculation involving electrode potentials is required to make accurate predictions in this area.\) Sodium is a soft metal that tarnishes within seconds of being exposed to the. 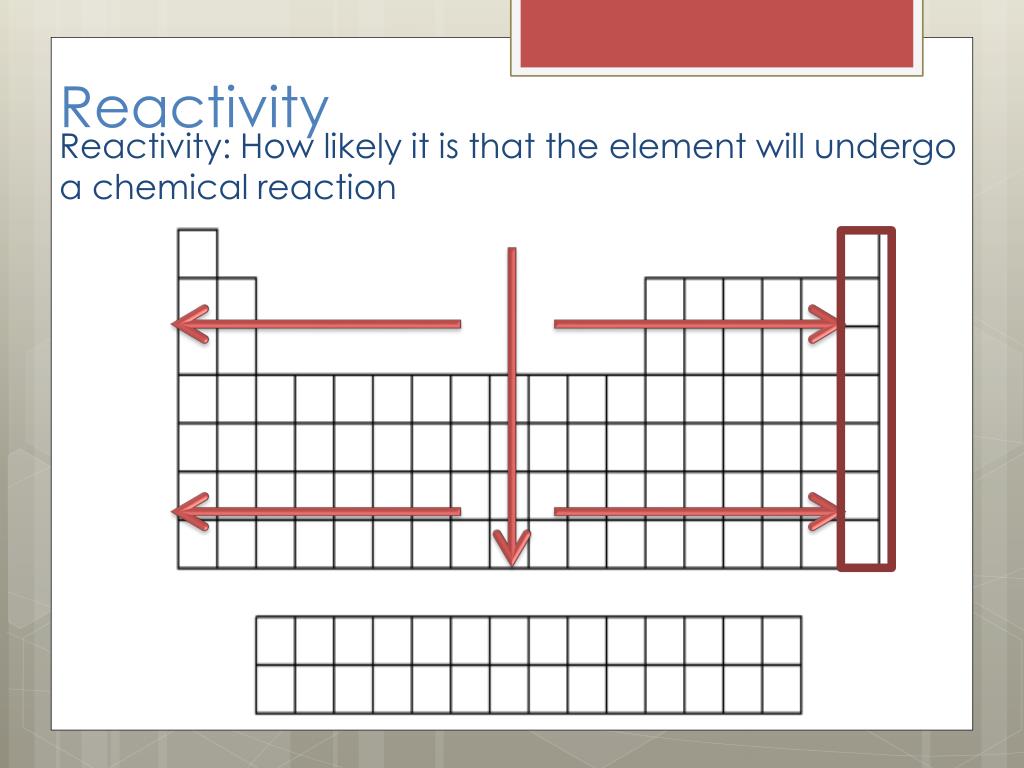
orange glow is like the colour of sodium street lighting and the spiked ‘flash’ symbol reflects the elements high reactivity. 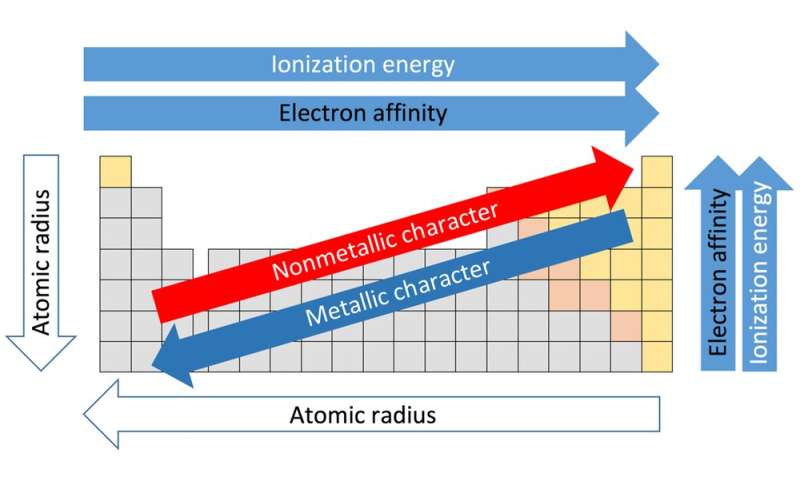
For example, calcium is quite reactive with water, whereas magnesium does not react with cold water but does displace hydrogen from steam. The atomic number of each element increases by one, reading from left to right. The boundary between the metals that react with water and those that don't is harder to spot. Those metals that can displace H + ions from acids are easily recognized by their position above H in the activity series. Less active metals like iron or zinc cannot displace hydrogen from water but do readily react with acids: Sodium is highly active and is able to displace hydrogen from water: It is important to distinguish between the displacement of hydrogen from an acid and hydrogen from water. 
However, silver cannot displace copper ions from solution. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
